Prof. Dr. med. Albert Bühlmann, University of Zürich
Published 1961 by J.R.Geigy S.A Switzerland «Der Weg in die Tiefe» in five bulletins
Experimental Decompression Research
If we summarise what has been said in the previous four bulletins, three main medical-physiological problems arise for diving:
- Oxygen poisoning
- The so-called deep intoxication and nitrogen narcosis
- Decompression
Oxygen poisoning can be safely avoided by reducing the oxygen concentration in the gas mixture. The deep intoxication could be explained as carbonic acid retention, in extreme cases as carbonic acid narcosis. The increasing gas weight at overpressure makes breathing so difficult that when individually varying limits are exceeded, there is no longer sufficient ventilation. Hypoventilation, however, means an increase in carbonic acid tension in the alveoli and in the blood with all secondary effects on the circulation and consciousness.
Deep intoxication is avoided if the load on the respiratory muscles is reduced, which is possible with artificial ventilation or by replacing the heavy nitrogen with specifically lighter gases such as helium and hydrogen. Corresponding calculations and investigations, and not least our successful deep diving experiments, prove that nitrogen under positive pressure does not have a specifically narcotic effect, as many diving experts claim.
There remains the third main problem, decompression.
There is not much point in descending to 200 meters for a few minutes, for example, if decompression takes 24 hours afterwards.
Deep diving can only gain practical importance if it is possible to shorten decompression times considerably.
The 4th bulletin discussed the physical principles of decompression, the gas saturation of the tissues depending on pressure and time as well as blood circulation. It was explained why the process of decompression must follow an asymptotic curve in time, as Haldane had already stated. His working model with a critical supersaturation factor of 1.8 for nitrogen was based on the observation that one can return to the surface immediately from a depth of 10 meters, i.e. 2 atm total pressure, even after long and repeated dives.
Decompression research begins with empiricism and has remained so for the most part. All modifications and improvements to Haldane’s original tables take into account decades of practical diving experience and especially the analysis of numerous decompression accidents. The progress has thus also been achieved less in the laboratory than empirically. However, the introduction of ever larger safety margins, which undoubtedly results in a reduction of accidents, has practically led to ever longer decompression times for the most important dives. Decompression is thus carried out as if the critical supersaturation factor for nitrogen were not 1.8 but, for example, only 1.4; theory and practice no longer coincide, although Haldane’s observation was undoubtedly correct and has been confirmed at all times. With the introduction of some new terms and a mathematical analysis of the problem, as carried out by H. Keller, theory and practice can be reunited, but we will not go into this in detail here.
Our new decompression method with its drastic shortening of resurfacing times now seems to contradict all tables and experience.
New decompression tables must therefore be developed for this method. The previous empirical way and the utilisation of accidents would lead much too slowly to the goal. Experiments that allow a maximum of statements with a minimum of risk promise much more success. Fundamental detailed investigations, such as gas dissolution in various body tissues under overpressure, require a specialised laboratory with the appropriate means, which are not available. There are not only affective reasons against animal experiments, but also great practical difficulties. How do you get the test animals to equalise pressure with the sinuses when the pressure rises?
Without pressure equalisation, severe pain, perhaps even fractures, would result. The simplest experiment is therefore with the diver himself. All the problems of interest here can be studied in the hyperbaric chamber. However, a hyperbaric chamber that would allow analogous conditions to the diving depths of 200 metres and more that we are interested in would require a great deal of constructional and material effort and, given today’s delivery times, a considerable waiting period.

The nearest efficient hyperbaric chamber is at the French navy in Toulon, about 800 km from Zurich, which does not exactly make experimentation any easier, despite the obliging nature of the French authorities. Our previous diving experiments in the water and in the hyperbaric chamber in Toulon (2nd Bulletin) involved very rapid descents followed by immediate ascent. However, the value of the method was only proven by the possibility of remaining at these depths for some time, doing work and still resurfacing in a relatively short time. The practical-minded Americans therefore demanded a stay of 10 minutes at a depth of 200 meters with physical work for a demonstration.
In order to carry out such a demonstration, preliminary tests must of course be made. A diving accident showed us the way to make a virtue out of necessity. In the summer of 1960, a professional diver suffered a typical decompression accident in a Swiss reservoir at an altitude of about 2000 meters. Decompression was carried out according to regulations, and yet dangerous bubbles formed. Why?
It had not been taken into account that the decompression tables apply to sea level, i.e. a normal pressure of 1 atm. At 1900 meters, however, the air pressure is only around 600 instead of 760 Torr, i.e. only 0.79 atm. A water depth of 10 meters naturally corresponds to an atmosphere even at this altitude, since the weight of the water does not decrease.
However, the decompression to the surface, to the current pressure of only 0.79 instead of 1 atm, was too abrupt, so that in some tissues the critical nitrogen supersaturation was exceeded and gas bubbles were released. In this case, decompression according to the table would have had to be carried out up to 2 meters below the water, where the pressure would then have just corresponded to 1 atm.
From 2 meters water depth to the surface, an additional decompression stage would have been necessary.
For decompression, as for pressure equalisation with the paranasal sinuses, it is not the absolute pressure that matters at all, but only the pressure differences. In the 4th Bulletin it was mentioned that decompression problems also arise for flying, because it does not matter whether the organism is exposed to a pressure drop from 2 to 1 or from 1 to 0.5 atm.
For experimental decompression tests, a large and expensive hyperbaric chamber is therefore not necessary at all. It is sufficient to combine a simple overpressure chamber with a negative pressure chamber. If, for example, the pressure is reduced to 0.2 atm, which corresponds to an altitude of 12,000 meters, and then allowed to rise again to 1 atm, the result of the fivefold increase in pressure in terms of additional gas absorption by the tissues is the same as for a dive to 40 meters depth. For people living permanently at an altitude of 12000 meters, a visit to the earth’s surface would have the same significance as a dive 40 meters below water level. If we increase the pressure to 4.2 atm, for example, we get a factor of 21 from 0.2 to 4.2, and thus the same conditions as a dive to 200 m. The return from 4.2 to 0.2 atm requires absolutely the same decompression ceremony as from 21 to 1 atm.
It was only a small step from this consideration to practical realisation. A small hyperbaric chamber has been available to us since 1959, and the combination with a vacuum pump offered no difficulties. All planned dives can be tested in this way with regard to decompression. This has some practical advantages. The usual decompression accidents usually occur during the last stages and after surfacing, in the negative pressure range for example between 1.0 and 0.2 atm.
The most effective treatment for bubble formation is always immediate recompression.
In the water, as in the hyperbaric chamber, the pressure must be increased or the dive must be made deeper so that decompression can be started again. In the negative pressure area, opening the chamber, i.e. the normal air pressure, is sufficient for a sufficient recompression. The explorant can get out of the chamber, there are no difficulties for transport and medical interventions. The return to normal pressure means optimal recompression without the need for subsequent decompression.
One disadvantage of the method is that the nitrogen content of the tissues, a total of about 700 ml at 1 atm, has to be adjusted to the initial pressure of 0.2 atm, i.e. reduced to about 140 ml. The explorer does not have to stay at 0.2 atm for hours, it is sufficient to breathe pure oxygen, for example in an oxygen tent on the surface for a few hours, because this also «washes» the nitrogen out of the tissues, as was explained in the 4th Bulletin. At 0.2 atm pure oxygen must of course be breathed, with air there would be severe hypoxia.
However, this does not falsify the conditions because oxygen can also be breathed at the surface at the beginning of a proper dive. Oxygen reduction at depth can also be carried out in the same way as on the real dive. If, for example, we only need 5% oxygen at a depth of 200 meters to avoid a dangerously high partial pressure of oxygen, this 5% is also sufficient for the fictitious dive at a pressure of 4.2 atm, as this just results in a normal partial pressure of oxygen of 150 Torr in the inhaled air. The fictitious dive can be carried out with the same gas mixtures as the real experiment. After several preliminary technical tests, we tested two representative fictitious dives in this way. In the first experiment, after a short descent to 265 meters, a stay of 20 minutes at a fictitious depth of 215 meters was observed. The subsequent decompression to 0.2 atm took place in 2 hours and 20 minutes (Fig.1).
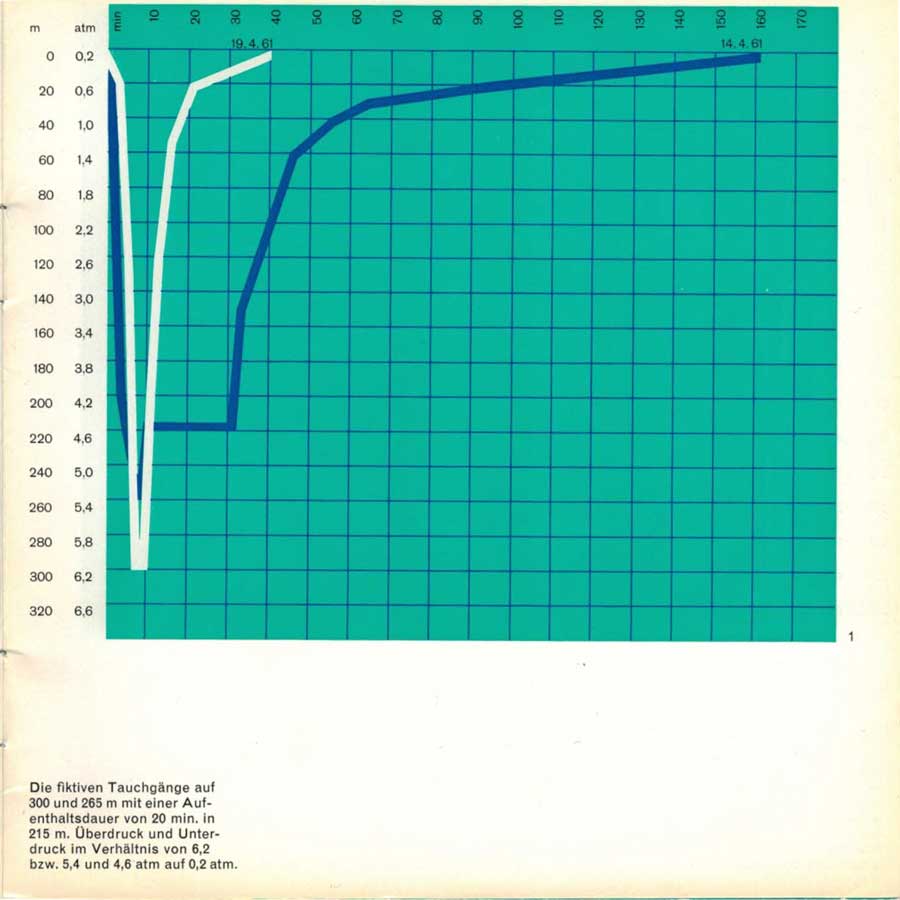
Overpressure and underpressure in the ratio of 6.2 and 5.4 and 4.6 atm to 0.2 atm respectively.
In order to find out whether damage might still occur later during this decompression time, H. Keller wanted to stay at 0.2 atm for another 30 minutes. After 15 minutes he began to experience slight pain in one knee joint, i.e. typical «bends”. The pressure was then increased to 1 at these complaints disappeared immediately. This proves that it really was a case of bubbles forming and not a coincidence. The test showed that the calculated decompression time for this dive was not quite sufficient to avoid late damage and must be corrected accordingly. In the second test, a fictitious descent to 300 meters was carried out with an immediately subsequent ascent. The decompression time was only 30 minutes. The difference in decompression times between the two experiments clearly shows the importance of the duration of stay.
With this combination of an overpressure and underpressure chamber, the most important decompression problems can be dealt with easily and relatively cheaply, because it is not the absolute pressure that is important, but only the pressure differences. However, as explained, this only applies to decompression. Where the absolute pressure is essential, as for example for the gas weight, which plays such an important role in hindering breathing at depth, only the effective diving test in the water and in the hyperbaric chamber with equivalent pressure can decide on the advantages and disadvantages of a diving method. But since decompression is the most difficult diving problem, the new test method, which to our knowledge has never been used before, can be considered experimental progress.
After these experiments with fictitious dives, we were immediately able to carry out a similar programme with real dives in the hyperbaric chamber of the French Navy in Toulon.
The descent to 300 meters on 25 April 1961 had a sensational effect as a new «world record».
For the experts, however, the bigger surprise was that we were able to demonstrate the following day how it is also possible with the new method to stay at greater depths for several minutes, to work physically and yet to return to the surface in a relatively short time (Fig.2).
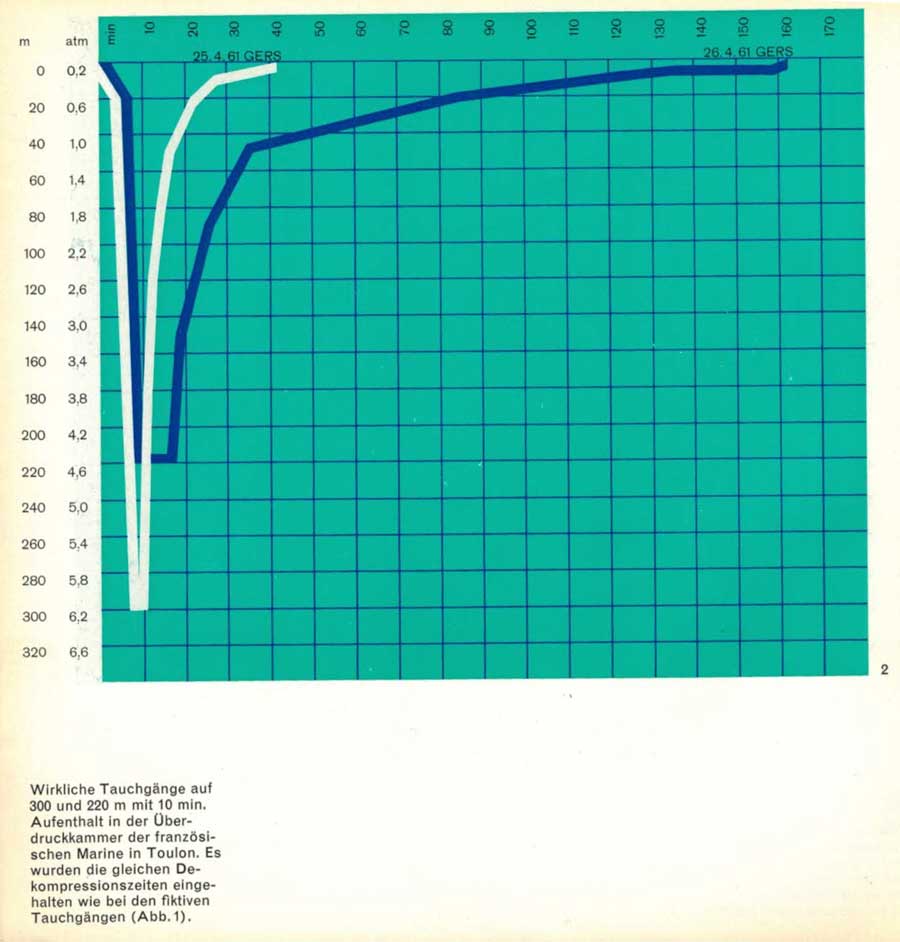
The same decompression times were respected as in the virtual dives.
Even though we did not doubt the correctness of our method in the least and therefore did not see any particular dangers on the medical-physiological side, possible technical deficiencies of the complicated apparatus and coordination errors within the operating team meant a risk.

The American doctors and naval officers present seemed very impressed by the preparations and the success of these experiments. We Swiss were naturally proud of this new success, but also full of gratitude and admiration for the French authorities and naval officers who, despite the simultaneous turbulent events in Algiers and their repercussions on Toulon, did everything they could to contribute to the success of the trials.





